2026-01-23
An Introduction to the Actinium-225 Nuclide and the ACTION-1 Study
Alpha-emitting radiopharmaceuticals utilize radionuclides that emit alpha particles, such as Radium-223 ([²²³Ra]), Actinium-225 ([²²⁵Ac]), Thorium-227 ([²²⁷Th]), Astatine-211 ([²¹¹At]), Lead-212 ([²¹²Pb]), and Bismuth-213 ([²¹³Bi]). Alpha particles are characterized by high Linear Energy Transfer (LET), strong ionizing radiation effects, high cytotoxic efficiency, and a short path length. They typically penetrate approximately 40–100 μm in tissue (less than 10 cell diameters). The high LET of alpha particles induces double-strand breaks in the DNA of tumor cell nuclei, leading to apoptosis. Moreover, due to their high potency, only a low radioactive dose is required, as merely a few alpha particle traversals through the cell nucleus are sufficient to kill the cell.
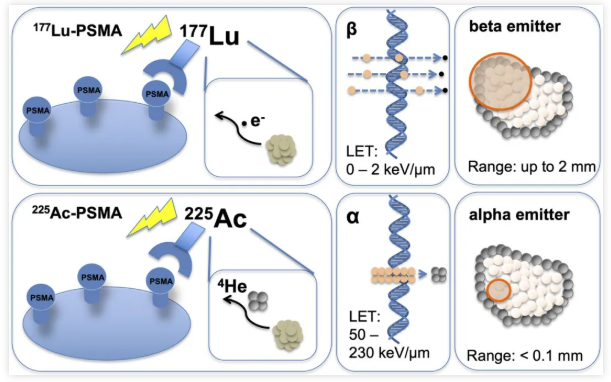
Comparison of the Radiobiological Effects between ²²⁵Ac-PSMA and ¹⁷⁷Lu-PSMA
Actinium-225 (²²⁵Ac), with a half-life of 9.92 days, has demonstrated remarkable therapeutic efficacy in both preclinical and clinical studies. This potency is primarily attributed to the emission of four alpha particles during its decay chain. However, ²²⁵Ac itself does not emit gamma rays suitable for direct imaging. Consequently, its biodistribution and imaging are typically reliant on gamma emissions from daughter radionuclides in its decay chain, specifically Francium-221 (²²¹Fr; half-life: 4.80 min; 218 keV; 11.4%) and Bismuth-213 (²¹³Bi; half-life: 45.6 min; 440 keV; 25.9%). The detection of ²²¹Fr and ²¹³Bi occurs following successive alpha decays, a process that may introduce certain inaccuracies due to daughter recoil and potential dissociation of the radiopharmaceutical conjugate. Therefore, the ability to directly and quantitatively detect actinium isotopes in vivo is crucial for optimizing preclinical radiopharmaceutical development and clinical treatment planning.
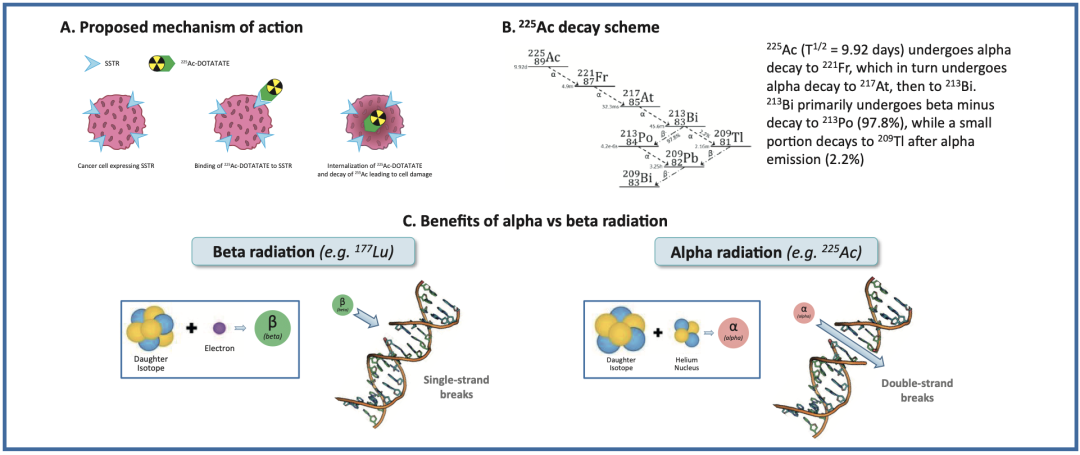 RYZ101 (²²⁵Ac-DOTATATE): A Radiopharmaceutical for Radionuclide Therapy
RYZ101 (²²⁵Ac-DOTATATE): A Radiopharmaceutical for Radionuclide Therapy
RYZ101 (²²⁵Ac-DOTATATE) is an alpha-particle emitting radiopharmaceutical currently under development for the treatment of solid tumors expressing somatostatin receptor subtype 2 (SSTR2+). The ACTION-1 (NCT05477576) trial is a global, randomized, controlled, open-label, two-stage Phase 1b/3 study. It aims to compare the efficacy of RYZ101 against standard of care in patients with inoperable, advanced, SSTR2+, well-differentiated gastroenteropancreatic neuroendocrine tumors (GEP-NETs) whose disease has progressed following prior therapy with lutetium-177 (¹⁷⁷Lu)-labeled somatostatin analogs.
Within the Phase 1b portion of the ACTION-1 trial, a dosimetry sub-study was conducted for the first time. This sub-study was designed to evaluate the feasibility of acquiring ²²⁵Ac imaging data to estimate the absorbed dose of RYZ101 to critical organs (primary objective) and tumors (secondary objective). The findings, presented at the JNM meeting in June 2023, confirmed the feasibility of ²²⁵Ac imaging and the acquisition of quantitative data required for dosimetry.
Within the Phase 1b portion of the ACTION-1 trial, the primary objective was to determine the Recommended Phase 3 Dose (RP3D) of RYZ101. To ensure at least five evaluable patients, eight patients were enrolled in the dosimetry sub-study.
Dosimetry calculations were performed for Cycles 1 and 4 using SPECT/CT imaging, acquired at 4 ± 1 hours, 24 ± 2 hours, and 168 ± 24 hours post-injection. During the ²²⁵Ac-labeled SPECT/CT scans, high-energy collimators were employed along with corresponding uniformity correction maps. Data acquisition was conducted across three specific energy windows (92 keV, 218 keV, and 440 keV, with window widths of 25%, 20%, and 20%, respectively).
By applying a dual-radionuclide quantitative SPECT reconstruction technique, distribution images of both ²²¹Fr and ²¹³Bi were obtained, with compensation for potential crosstalk and other physical factors affecting the image results. Precise quantitative analysis of radioactivity was performed for the liver, kidneys, spleen, red marrow (L3–L5 vertebrae), and selected tumors.
The absorbed doses for target organs and lesions were calculated using the S-value method recommended by the Medical Internal Radiation Dose (MIRD) Committee, in conjunction with the 3D-RD-S tool within the Rapid software package. Overall, the study revealed for the first time that ²¹³Bi predominantly remains associated with the targeting vector (DOTATATE), with only a minor fraction accumulating in the kidneys. Preliminary dosimetry results from ACTION-1 support a favorable tumor-to-background ratio for RYZ101 in treating SSTR+ GEP-NETs. A fixed dose of 10.2 MBq has been established as the RP3D for Phase 3 clinical trials.
The objective of radionuclide dosimetry is to estimate the absorbed dose in normal tissues and tumors, thereby predicting the biological effects of radiation. When performing dosimetry for alpha-emitting radiopharmaceuticals, although the estimated absorbed dose from alpha particles can be multiplied by a Relative Biological Effectiveness (RBE) factor, it is also necessary to consider the impact of microscopic distribution differences of the radionuclide on the discrepancy between predicted and observed toxicities.
Given that current technology cannot precisely measure the activity of alpha-emitting radiopharmaceuticals and their decay daughters in vivo, surrogate methods can be employed to assess radiation exposure to tumors and normal organs until accurate quantitative techniques become available. These methods can guide the optimization of dose selection based on dose-response relationships, as well as clinical efficacy and safety data.
For alpha-emitting radiopharmaceuticals that cannot be directly imaged, it is recommended to utilize established imaging techniques to detect the radiolabeled targeting vector. For instance, single-photon emitting radionuclides detectable by SPECT (such as Indium-111 [¹¹¹In] or Technetium-99m [⁹⁹ᵐTc]), or positron-emitting radionuclides detectable by PET (such as Zirconium-89 [⁸⁹Zr], Gallium-68 [⁶⁸Ga], Copper-64 [⁶⁴Cu], or Fluorine-18 [¹⁸F]) can be used. However, due to potential differences in energy deposition, dose rate, and pharmacokinetics between the surrogate and the therapeutic radionuclide, these methods serve only as auxiliary tools for estimating alpha-particle radiation exposure. The actual distribution of the alpha-emitting radiopharmaceutical must be confirmed through further in vivo studies.
Radiation dosimetry exhibits high variability due to underlying patient conditions and individual differences, which limits its ability to predict individual toxicity and/or efficacy. Therefore, radiation dosimetry should serve as supplementary information for evaluating clinical safety and efficacy within dose-escalation studies. In clinical trials, decisions regarding dose escalation and individual patient management should be based on clinical safety assessments rather than relying solely on planned radiation absorbed dose limits.
[²²⁵Ac]Ac as a Surrogate Imaging Agent: [²²⁶Ac]Ac
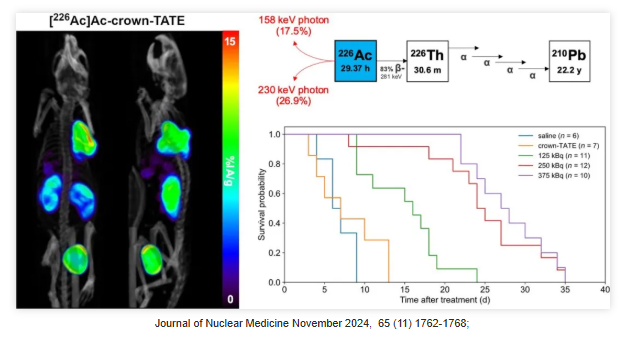
Actinium-225 (²²⁵Ac) has garnered significant attention as a therapeutic radioisotope due to the alpha particles emitted during its decay chain. However, the lack of directly imageable gamma radiation limits the direct observation of its in vivo distribution. Consequently, surrogate imaging agents, such as Actinium-226 (²²⁶Ac), are crucial for evaluating the in vivo distribution of ²²⁵Ac, optimizing treatment planning, monitoring therapeutic efficacy, and ensuring both safety and personalization.
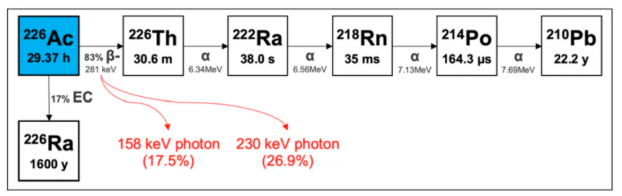
²²⁶Ac (half-life: 29.37 hours) is ideally suited as a "theranostic match" for ²²⁵Ac-labeled radiopharmaceuticals, as they share identical in vivo radiochemistry and pharmacokinetic properties. ²²⁶Ac can be imaged via SPECT using its 158 and 230 keV gamma emissions. Furthermore, due to the alpha decay of its daughter isotope ²²⁶Th, ²²⁶Ac can also be regarded as a standalone therapeutic isotope.
²²⁶Ac undergoes beta decay to form ²²⁶Th, which subsequently emits four high-energy alpha particles via very short-lived daughters, resulting in a cumulative energy of 27.7 MeV. Recently, a study published in the Journal of Nuclear Medicine (JNM) evaluated the feasibility of ²²⁶Ac (half-life: 29.37 hours) as a standalone therapeutic radionuclide and demonstrated its potential as a therapeutic agent.
The Decay Chain and Physical Properties of Actinium-226
The results revealed a high degree of consistency between quantitative SPECT imaging and ex vivo measurements for [²²⁶Ac]Ac-crown-TATE in vivo. This agent demonstrated high tumor uptake (exceeding 30% IA/g within 5 hours post-injection) and prolonged retention, delivering a mean absorbed dose of 222 mGy/kBq. Treatment with [²²⁶Ac]Ac-crown-TATE significantly suppressed tumor growth and prolonged survival in mice, without observed weight loss or toxic reactions throughout the study.
Overall, this study highlights the potential of ²²⁶Ac as a standalone therapeutic isotope for both diagnosis and therapy. It not only validates its diagnostic capability for dosimetry in matched ²²⁵Ac radiopharmaceuticals but also suggests that future research should further investigate maximum dosing and toxicity to fully exploit the therapeutic potential of ²²⁶Ac-based agents.
Such surrogate imaging agents provide critical dosimetric information, enabling researchers and clinicians to gain a deeper understanding of ²²⁵Ac biodistribution. This facilitates more precise tumor ablation in Targeted Alpha Therapy (TAT) while minimizing damage to surrounding normal tissues. Furthermore, the application of surrogate imaging agents is of great significance for novel drug development and the establishment of theranostic pairs, thereby further advancing the field of nuclear medicine and offering patients more effective and safer treatment options.
[²²⁵Ac]Ac as a Surrogate Imaging Agent: [¹³⁴Ce]Ce
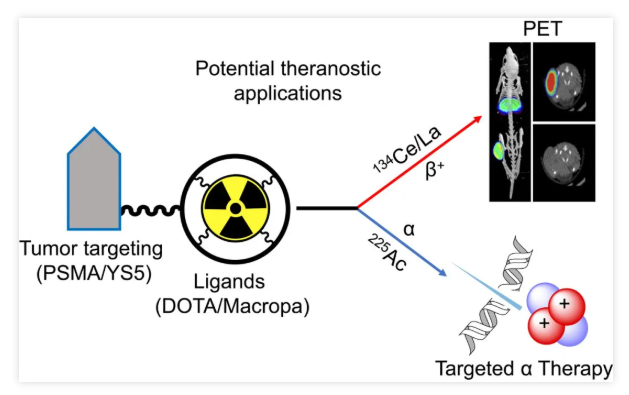
In July 2023, a study published in the Journal of Nuclear Medicine (JNM) comprehensively evaluated the potential of Cerium-134 (¹³⁴Ce) and Lanthanum-134 (¹³⁴La) as a Positron Emission Tomography (PET) theranostic pair for Actinium-225 (²²⁵Ac)-based alpha-radionuclide therapy. The research achieved efficient radiolabeling of ¹³⁴Ce using DOTA and MACROPA chelators, applying this to assess the in vivo pharmacokinetic properties of the prostate cancer imaging agents PSMA-617 and MACROPA-PEG4-YS5, and compared these with their corresponding ²²⁵Ac analogs.
The findings demonstrated that ¹³⁴Ce shares similar chemical and pharmacokinetic characteristics with ²²⁵Ac. The ¹³⁴Ce/¹³⁴La pair is thus a promising candidate for PET imaging surrogates in ²²⁵Ac-based radioligand therapy. Specifically, ¹³⁴Ce-MACROPA.NH2 and ¹³⁴Ce-DOTA exhibited high in vivo stability and biodistribution profiles similar to their ²²⁵Ac counterparts. Furthermore, PET imaging and biodistribution results of ¹³⁴Ce-PSMA-617 and ¹³⁴Ce-MACROPA-PEG4-YS5 in prostate cancer models support the potential of the ¹³⁴Ce/¹³⁴La pair as an imaging surrogate for ²²⁵Ac.
In June 2024, another study published in the European Journal of Nuclear Medicine and Molecular Imaging further conducted a direct comparison between Actinium-225 (²²⁵Ac) and Cerium-134 (¹³⁴Ce) labeled tracers, evaluating their potential as a matched theranostic pair. The study employed both fast-internalizing and slow-internalizing models to assess their in vivo similarity, daughter nuclide redistribution, and clinical translatability. The PSMA-617 precursor and the macrocyclic-based MCP-PEG8-Tz were radiolabeled with ²²⁵Ac and ¹³⁴Ce, respectively, and compared both in vitro and in vivo using standard (radio)chemical methodologies.
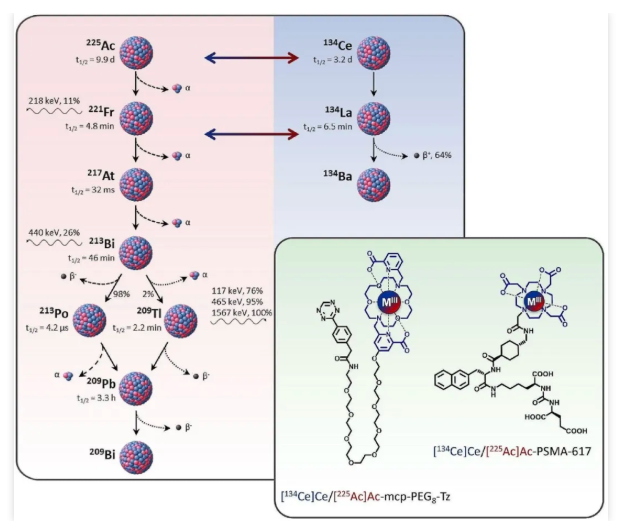
The in vitro and in vivo results demonstrated that tracers labeled with ²²⁵Ac and ¹³⁴Ce exhibit similar pharmacokinetic properties, thereby validating their potential as a matched theranostic pair. However, PET imaging analysis of ¹³⁴Ce-labeled precursors revealed that quantitative accuracy is highly dependent on the tracer's internalization efficiency—a phenomenon attributed to the in vivo redistribution of ¹³⁴La, the daughter nuclide generated from ¹³⁴Ce decay.
Consequently, radiotracers utilizing fast-internalizing vectors, such as PSMA-617, are well-matched with the ¹³⁴Ce/¹³⁴La theranostic pair. In contrast, ²²⁵Ac-labeled tracers with slow internalization kinetics are difficult to quantify accurately via ¹³⁴Ce PET imaging. In the case of slow-internalizing vectors, ¹³⁴Ce may be insufficient as an ideal surrogate for ²²⁵Ac, as the in vivo redistribution of ¹³⁴La could lead to an underestimation of tumor uptake.
Nonetheless, this characteristic offers the possibility of non-invasively monitoring the redistribution of ²²⁵Ac daughters. In future research, this could aid in the in-depth investigation of tracer internalization processes, receptor trafficking mechanisms, and alterations in the tumor microenvironment. Therefore, combining fast-internalizing radiotracers with a delayed imaging timepoint (e.g., 24 hours post-injection) is key to optimizing this theranostic strategy.
Conclusion
Actinium-225 (²²⁵Ac), as a highly promising alpha-particle-emitting radionuclide, has attracted significant attention in the field of targeted radionuclide therapy for tumors. It offers innovative therapeutic avenues for various malignancies due to its superior energy transfer efficiency, suitable half-life, short particle range, and excellent coordination properties.
Nonetheless, the clinical application of ²²⁵Ac still faces multiple challenges. These include limitations in radionuclide supply, the development of efficient labeling techniques and the screening of suitable chelators, enhancement of targeting precision, and mitigation of radiation damage to surrounding normal tissues. Despite these hurdles, with continuous innovations in dosimetry methodologies, precursor design, and radiopharmaceutical delivery technologies, alpha-radionuclide therapy holds great promise to evolve into a highly effective modality, significantly improving both the efficacy and safety of tumor treatment.