2026-01-23
The B7-H3 ADC Experience
In 2020, Dr. Tongtong Xue co-founded Yilian Biology alongside a team of seasoned industry experts. As an innovative biotech company focused on the research and development of tumor-targeted therapeutics, Yilian Biology is dedicated to advancing next-generation oncology drugs with breakthrough efficacy. The company places its core emphasis on the continuous innovation and iteration of cutting-edge ADC (Antibody-Drug Conjugate) technologies.

In March 2025, Yilian Biology announced a breakthrough in the Phase I clinical trial of YL201, its B7H3-targeting ADC candidate for advanced solid tumors. The findings were published in the peer-reviewed journal Nature Medicine under the title "A B7H3-targeting antibody–drug conjugate in advanced solid tumors: a phase 1/1b trial." The corresponding authors include Professor Li Zhang, Professor Hongyun Zhao from Sun Yat-sen University Cancer Center, and Dr. Tongtong Xue, CEO of Yilian Biology.
This global study, conducted in collaboration with 54 leading oncology centers, enrolled 312 patients with advanced solid tumors who had failed multiple lines of prior therapy. The results demonstrated breakthrough efficacy data in extensive small cell lung cancer (ES-SCLC), nasopharyngeal carcinoma (NPC), lung lymphoepithelioma-like carcinoma (LELC), and non-small cell lung cancer (NSCLC). The drug exhibited objective response rates (ORR) and disease control rates (DCR) that were significantly superior to existing standard-of-care therapies. Notably, YL201 displayed an excellent safety profile, laying the foundation for subsequent pivotal Phase III clinical trials.
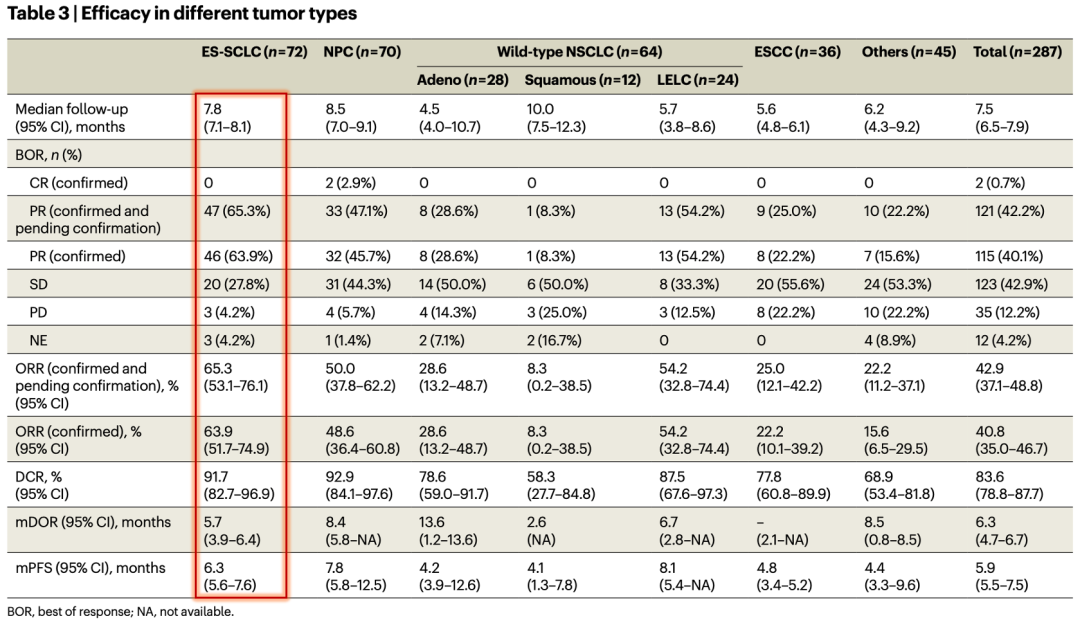
Among 72 evaluable patients with extensive-stage small cell lung cancer (ES-SCLC), YL201 demonstrated breakthrough efficacy: the objective response rate (ORR) reached 63.9%, the disease control rate (DCR) was 91.7%, and the median progression-free survival (mPFS) was 6.3 months. These results represent nearly a twofold improvement compared to existing second-line standard-of-care therapies—topotecan (ORR ≈ 24%) and lurbinectedin (ORR ≈ 35%)—significantly reshaping the landscape of later-line treatment for ES-SCLC.
Based on these promising data, Yilian Biology is accelerating the pivotal Phase III clinical development of YL201 in the SCLC population (NCT06612151). The company has initiated a multicenter, randomized, open-label, active-comparator controlled Phase III study titled "A Phase III Study to Evaluate the Efficacy and Safety of YL201 Injection Versus Topotecan Hydrochloride Injection in Patients with Recurrent Small Cell Lung Cancer."
 This study revealed for the first time that B7-H3 protein expression—whether membranous or in soluble form—shows no significant correlation with the clinical efficacy of YL201. Consequently, YL201 may not require pre-screening of patients with high B7-H3 expression via immunohistochemistry (IHC) or liquid biopsy. This finding is expected to significantly streamline the clinical application pathway and broaden the population of potential beneficiaries.
This study revealed for the first time that B7-H3 protein expression—whether membranous or in soluble form—shows no significant correlation with the clinical efficacy of YL201. Consequently, YL201 may not require pre-screening of patients with high B7-H3 expression via immunohistochemistry (IHC) or liquid biopsy. This finding is expected to significantly streamline the clinical application pathway and broaden the population of potential beneficiaries.
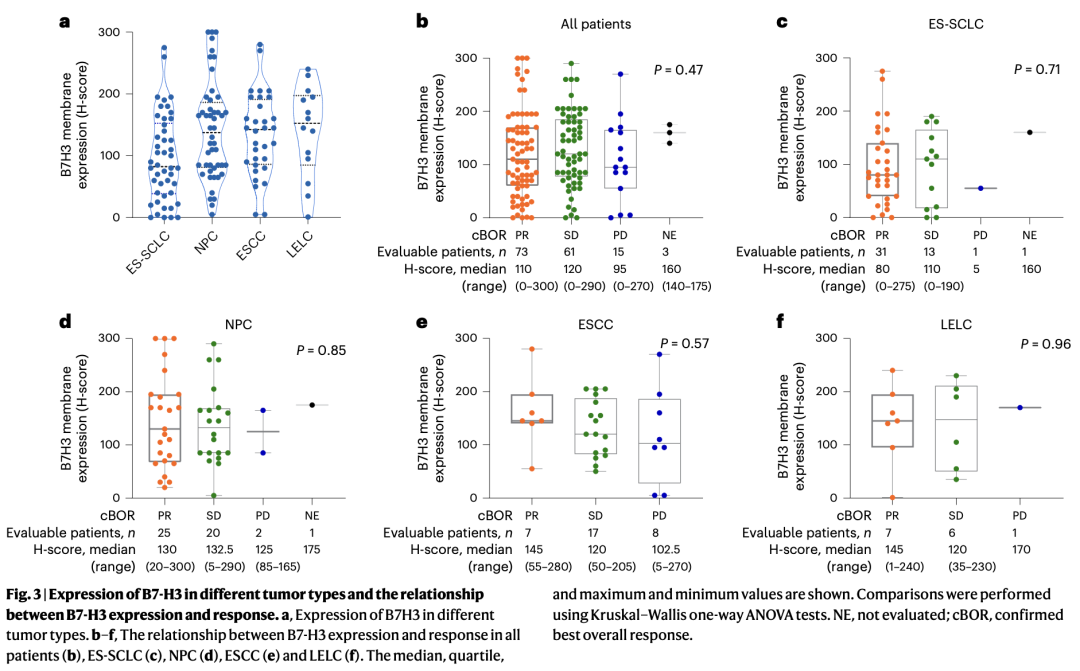
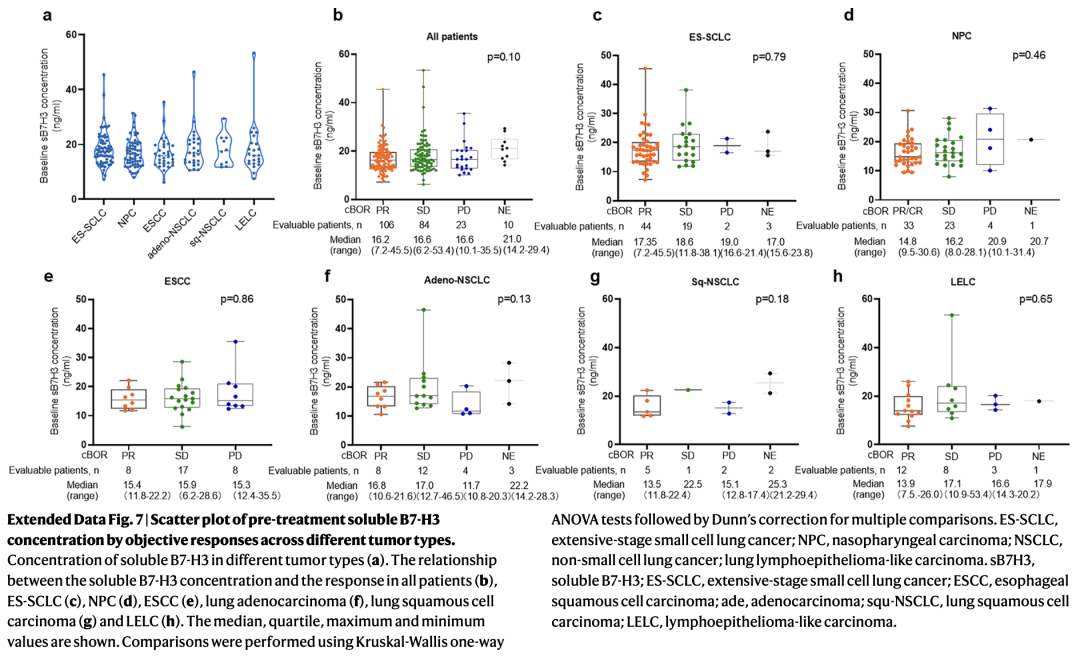
Detailed data are as follows: Investigators assessed membranous B7-H3 protein expression levels (measured by H-score, range 0–300) in tumor tissue samples from 152 patients using immunohistochemistry (IHC), while simultaneously measuring the concentration of soluble B7-H3 (sB7-H3) in patient plasma.
The results indicated that B7-H3 expression was detected in all tumor types enrolled in the study; however, the median H-scores varied significantly across different tumor histologies:
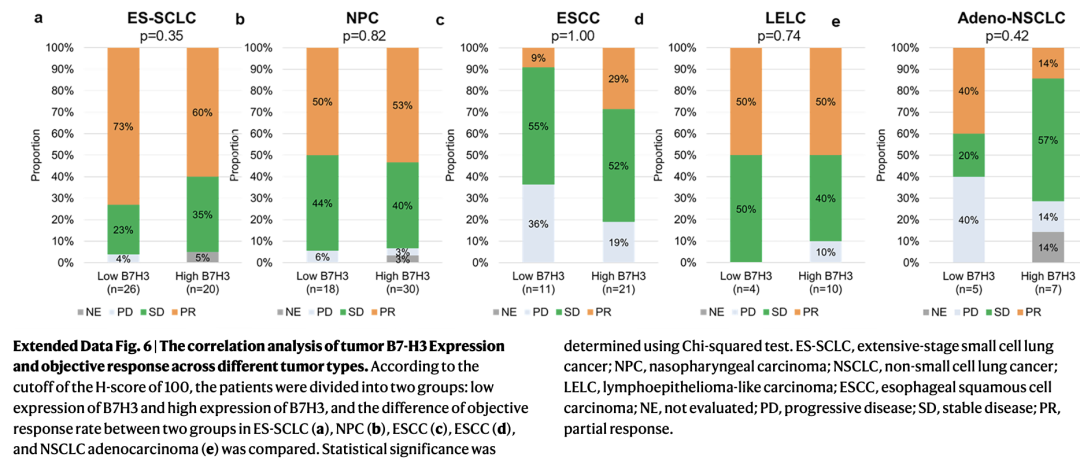
Although B7-H3 expression levels varied across different tumor types (e.g., highest in LELC and lowest in SCLC), the Kruskal-Wallis test comparing the median B7-H3 expression levels among different response groups showed no significant difference in H-scores between responders and non-responders (P-value did not reach the significance threshold). This indicates that membranous B7-H3 expression levels are not significantly correlated with the clinical efficacy of YL201 (ORR/DCR).
Furthermore, an analysis of soluble B7-H3 (sB7H3) concentrations in pre-treatment blood samples from 223 patients revealed that sB7H3 levels were not significantly associated with treatment outcomes (P > 0.05). This suggests that peripheral blood sB7H3 levels cannot serve as a predictive biomarker for YL201 efficacy.
The next phase of research is currently focused on developing highly specific radioactive diagnostic probes. These agents enable molecular-level visualization via PET/CT and SPECT/CT imaging. Examples include tracers targeting Trop2—a pan-cancer target generating significant momentum in the radiopharmaceutical field—and Nectin-4, where drug development is rapidly accelerating. These imaging tools hold promise for identifying optimal patient populations for ADC therapy via imaging-based target screening, while also providing molecular imaging assessment methods for selecting clinical indications for corresponding ADCs.
In my opinion, although existing studies indicate that the clinical efficacy of B7-H3-targeting ADCs is not significantly correlated with target expression levels, developing Targeted Radionuclide Therapy (TRT) remains clinically valuable. Unlike ADCs, which rely on the delivery of chemical toxins, radiopharmaceuticals induce physical tumor ablation via high-energy radiation. This radiation not only covers heterogeneous tumor regions but also disrupts the immunosuppressive microenvironment due to its penetration capability.
Furthermore, radioactive isotopes allow for the precise selection of target-positive patients via PET imaging prior to treatment and enable real-time monitoring of target occupancy and radiation dose distribution during therapy. This dynamic visualization capability allows TRT to overcome the limitations of traditional biomarker stratification, thereby opening new avenues for patients who do not respond to ADC treatments.
The Radiopharmaceutical: ⁸⁹Zr-DS-5573a
 In July 2018, Professor Andrew M. Scott—Head of the Department of Molecular Imaging and Therapy at Austin Health and Head of the Tumor Targeting Laboratory at the Olivia Newton-John Cancer Research Institute—served as a co-corresponding author on a research paper titled "Molecular imaging of T cell co-regulator factor B7-H3 with ⁸⁹Zr-DS-5573a," published in the journal Theranostics. The study reported the development of a novel molecular imaging probe, ⁸⁹Zr-DS-5573a, designed to target and detect the expression of the immune regulatory protein B7-H3 in tumors, exploring its potential for theranostic applications.
In July 2018, Professor Andrew M. Scott—Head of the Department of Molecular Imaging and Therapy at Austin Health and Head of the Tumor Targeting Laboratory at the Olivia Newton-John Cancer Research Institute—served as a co-corresponding author on a research paper titled "Molecular imaging of T cell co-regulator factor B7-H3 with ⁸⁹Zr-DS-5573a," published in the journal Theranostics. The study reported the development of a novel molecular imaging probe, ⁸⁹Zr-DS-5573a, designed to target and detect the expression of the immune regulatory protein B7-H3 in tumors, exploring its potential for theranostic applications.
ELISA assays confirmed that DS-5573a specifically binds to human B7-H3 without recognizing murine B7-H3. Lindmo analysis showed an initial immunoreactivity of 75.0±2.9% against B7-H3-positive MDA-MB-231 cells, compared to only 10.85±0.11% for B7-H3-negative CT26 cells. Further Scatchard analysis revealed a high affinity with an association constant (Ka) of 4.0×10⁹ M⁻¹ and approximately 87,000 binding sites per cell. Notably, the probe retained 57.2% of its immunoreactivity in human serum after 7 days.
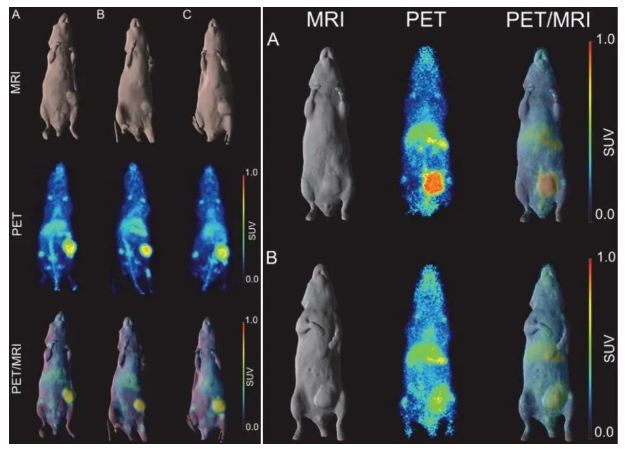
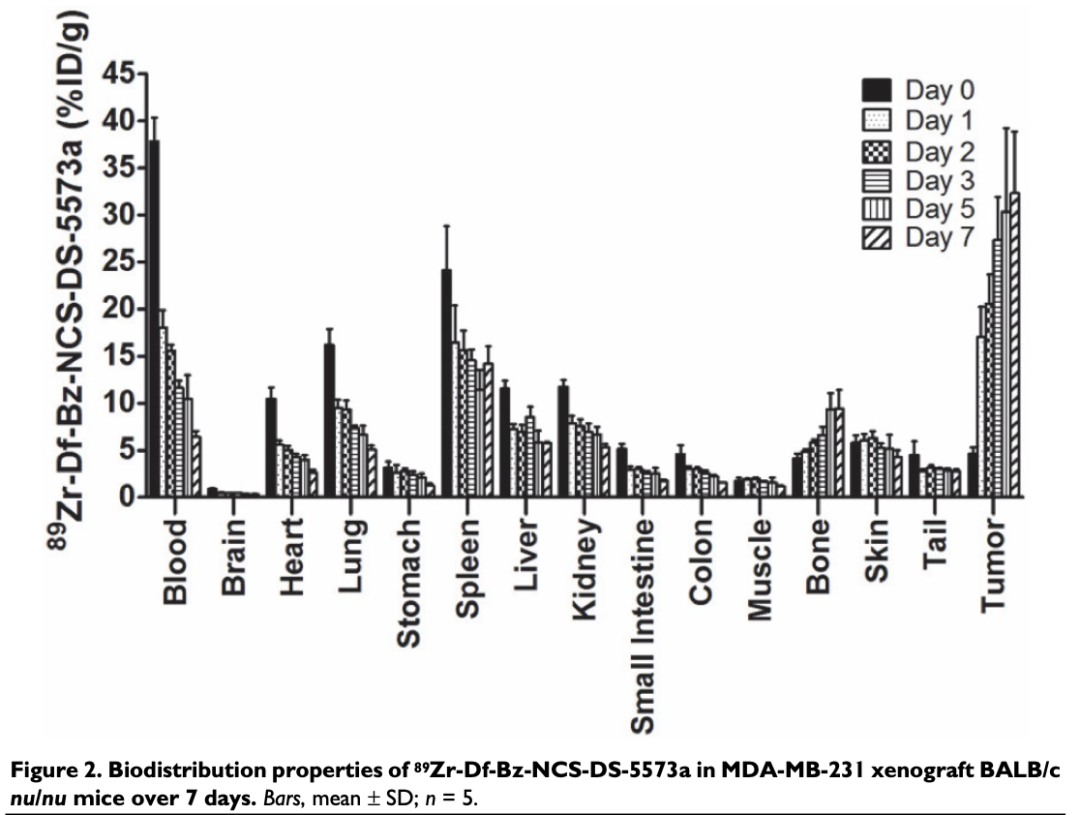
In vivo studies demonstrated a significant time-dependent increase in probe uptake within B7-H3-positive tumors, reaching 32.32±6.55% ID/g by day 7, with a tumor-to-blood ratio of 5.03±0.73. In contrast, B7-H3-negative CT26 tumors showed only 10.07% ID/g. PET/MRI imaging further confirmed its precise tumor localization capability.
In therapeutic experiments, a treatment dose of 3 mg/kg DS-5573a induced 89.8% tumor growth inhibition. When administering a diagnostic dose (5 μg) alongside a therapeutic dose (70 μg) of the antibody, tumor uptake of the probe decreased to 16.04±1.97% ID/g (versus 58.98±16.86% ID/g in the group without therapeutic antibody), confirming a receptor saturation effect caused by the therapy. This saturation state persisted for at least one week after the final treatment.
 In summary, the study demonstrates that ⁸⁹Zr-DS-5573a not only enables non-invasive quantification of tumor B7-H3 expression levels but also provides critical guidance for clinical dose optimization through dynamic monitoring of receptor occupancy. Exhibiting an excellent safety profile overall, this foundational work paved the way for the development of DS-7300 (Ifinatamab Deruxtecan)—the first B7-H3-targeting ADC to advance into Phase III clinical trials to date.
In summary, the study demonstrates that ⁸⁹Zr-DS-5573a not only enables non-invasive quantification of tumor B7-H3 expression levels but also provides critical guidance for clinical dose optimization through dynamic monitoring of receptor occupancy. Exhibiting an excellent safety profile overall, this foundational work paved the way for the development of DS-7300 (Ifinatamab Deruxtecan)—the first B7-H3-targeting ADC to advance into Phase III clinical trials to date.
[89Zr]Zr-Ab-1/2

In January 2025, Professor Orit Jacobson from the Molecular Imaging Program at the Center for Cancer Research, National Cancer Institute (NCI), part of the National Institutes of Health (NIH), served as the corresponding author of a research paper titled "Unraveling the dynamics of B7-H3-targeting therapeutic antibodies in cancer through PET imaging and antibody pharmacokinetics," published in the Journal of Controlled Release.
The study utilized PET imaging and antibody pharmacokinetic analysis to reveal the dynamic differences and underlying mechanisms of two B7-H3-targeting therapeutic antibodies (Ab-1 and Ab-2) within tumors.
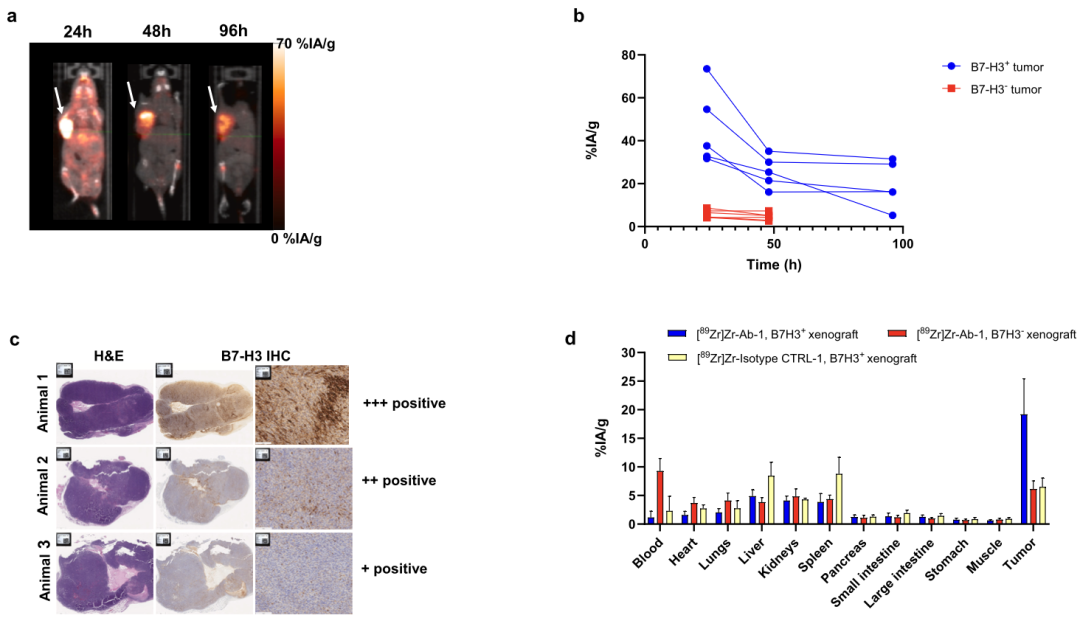
The study revealed that Ab-1 exhibits high affinity for both the 4Ig and 2Ig isoforms of B7-H3 (4Ig KD = 2 nM, 2Ig KD = 26 nM), achieving high tumor-specific uptake of up to 74% ID/g within 24 hours post-injection. However, its retention time was significantly shorter than expected, with tumor activity declining to 5–31% ID/g by 96 hours.
In-depth mechanistic analysis indicated that the rapid clearance of Ab-1 is closely associated with proteolytic degradation mediated by matrix metalloproteinases (MMPs)—particularly MMP9 and MMP2—within the tumor microenvironment. These enzymes cleave the 2Ig isoform bound to Ab-1, leading to the dissociation of the antibody-antigen complex. The resulting small fragments are subsequently released and excreted via urine.
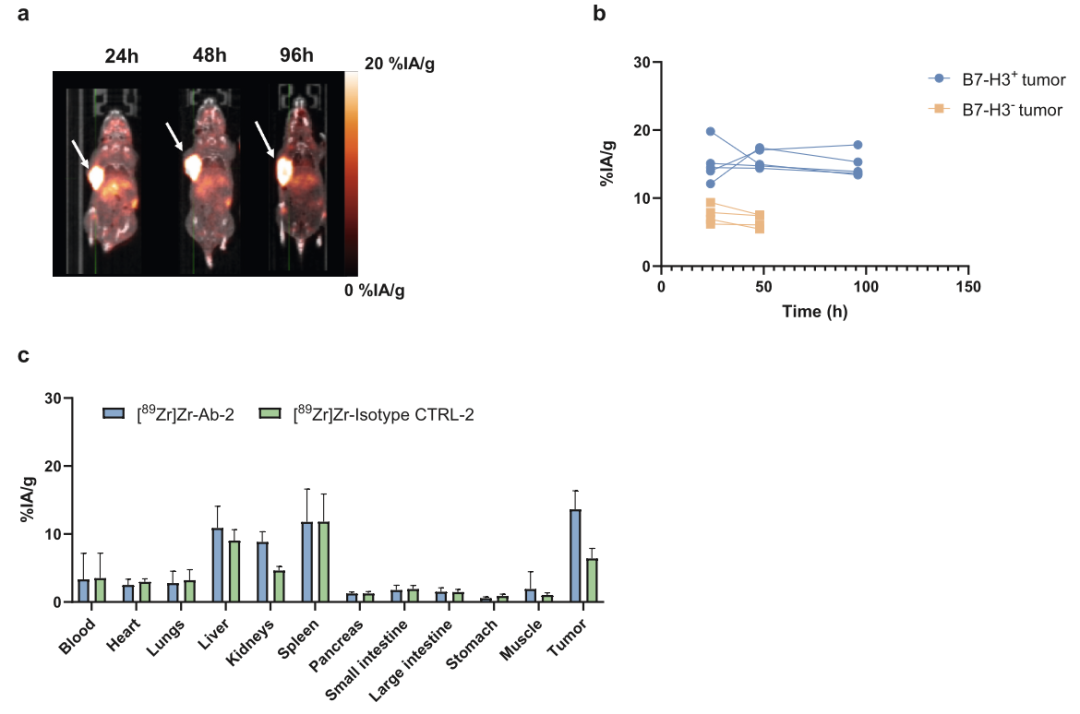 In contrast, Ab-2—which specifically binds only to the 4Ig isoform (KD = 3 nM)—demonstrated stable tumor retention (retaining approximately 80% of initial uptake at 96 hours). However, its overall uptake was lower than that of Ab-1, and it exhibited higher accumulation in the liver and spleen.
In contrast, Ab-2—which specifically binds only to the 4Ig isoform (KD = 3 nM)—demonstrated stable tumor retention (retaining approximately 80% of initial uptake at 96 hours). However, its overall uptake was lower than that of Ab-1, and it exhibited higher accumulation in the liver and spleen.
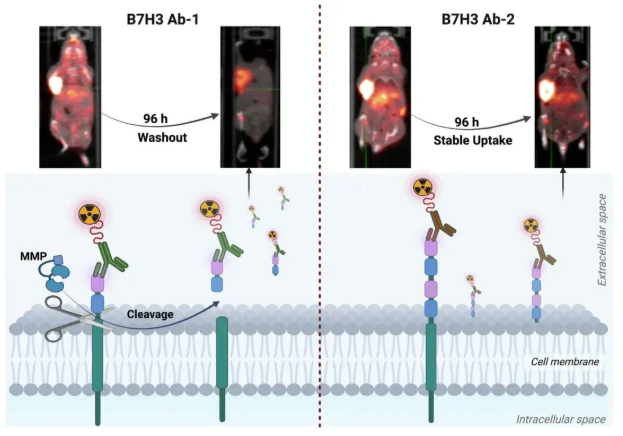
The study indicates that the tumor distribution and retention of B7-H3-targeting antibodies are co-regulated by antibody-antigen binding characteristics (isoform selectivity), the tumor microenvironment (protease activity), and antibody pharmacokinetics.
The high penetration and rapid clearance profile of Ab-1 make it suitable for fractionated radiotherapy or pre-targeted strategies employing short-half-life radionuclides (e.g., ²¹²Pb, ⁶⁷Cu). In contrast, the stable retention characteristics of Ab-2 are better suited for sustained radiation-induced cytotoxicity using long-half-life radionuclides (e.g., ⁹⁰Y).
The complementary properties of Ab-1 and Ab-2 suggest that personalized therapeutic regimens should be tailored based on both tumor biological features and the characteristics of the radionuclide used.
The Radiopharmaceutical: [¹⁷⁷Lu]Lu-FL801

AACR 2025: Dr. Fa Liu, Chief Scientific Officer of Fusion Pharmaceutical Co., presents two poster sessions.
Fusion Pharmaceuticals has developed the Clear-X™ linker technology, specifically designed to optimize the suboptimal biodistribution commonly observed in VHHs and other protein vectors of similar size, such as Affibodies and DARPins. Leveraging this Clear-X™ technology, Fusion has developed FL801, a radioligand targeting B7-H3.
In vitro assays demonstrated that FL801 exhibits sub-nanomolar affinity for B7-H3 (<1 nM). The radiolabeled conjugate, [¹⁷⁷Lu]Lu-FL801, achieved a high radiochemical purity (RCP) exceeding 98%, with a specific activity of 18.5 MBq/nmol. Furthermore, the compound remained stable for 5 days when stored in buffer at 4°C.
 Source: Poster presentation at AACR 2025 by Fusion Pharmaceuticals.
Source: Poster presentation at AACR 2025 by Fusion Pharmaceuticals.
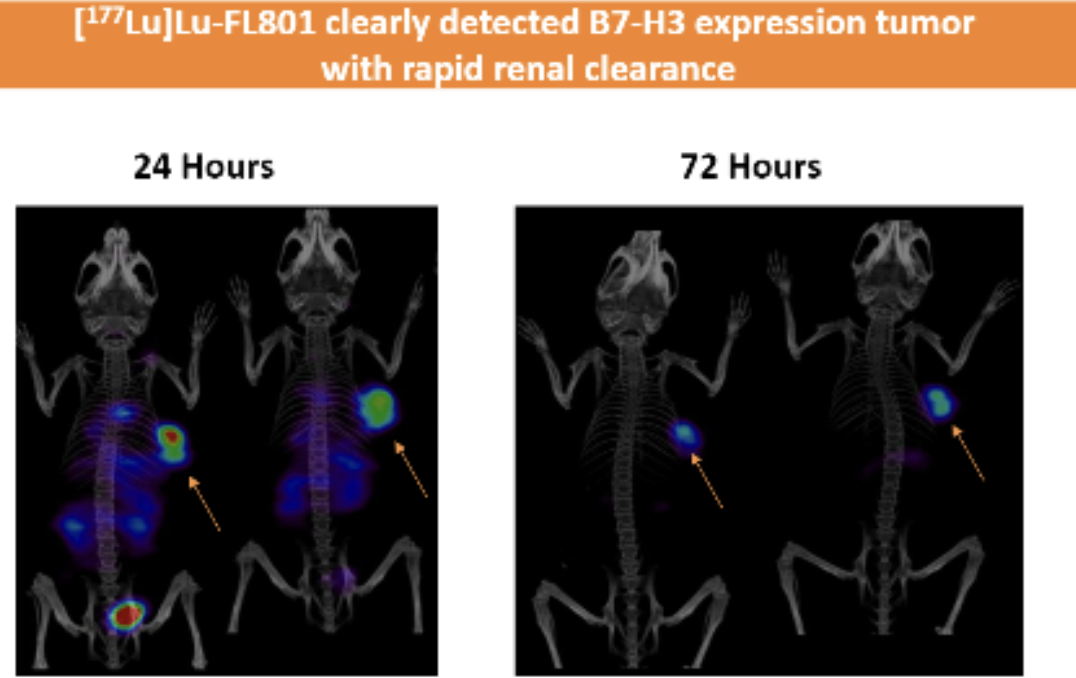
SPECT/CT imaging and ex vivo tissue counting analyses demonstrated that [¹⁷⁷Lu]Lu-FL801 exhibits favorable in vivo distribution characteristics in B7-H3-positive tumor-bearing mice.
Significant tumor uptake was observed:
This represents a substantial improvement compared to the unmodified parental VHH vector (without Clear-X™ modification), which showed a tumor uptake of only 2.3% ID/g and a T/K ratio <0.01.
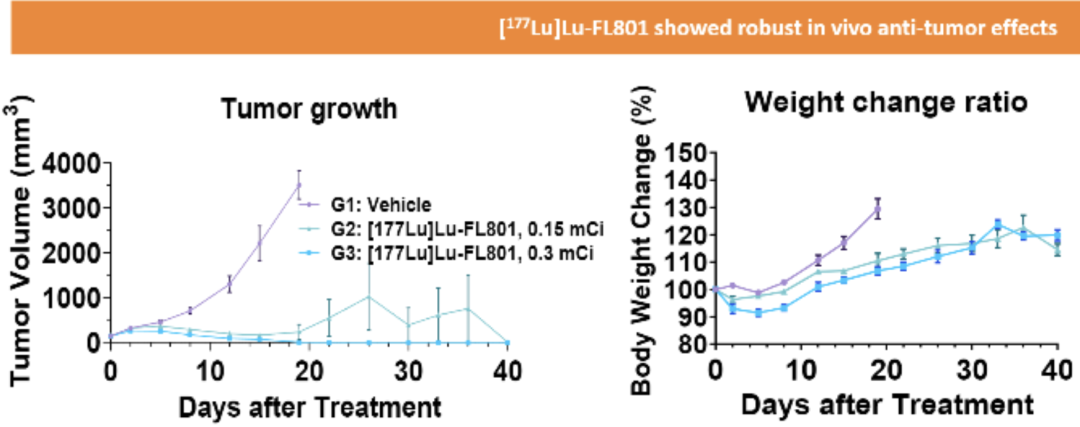
In tumor-bearing mouse models administered a single injection of 11.1 MBq [¹⁷⁷Lu]Lu-FL801, the treatment was well-tolerated, and all animals achieved complete tumor regression.
In conclusion, these preclinical data indicate that [¹⁷⁷Lu]Lu-FL801 holds promise as a potential therapeutic candidate for B7-H3-positive tumors, warranting its advancement into clinical studies. Furthermore, the Clear-X™ technology has successfully addressed the biodistribution challenges associated with small-protein-vector RDCs. This platform approach is readily extendable to other modalities such as Affibody and DARPins, paving the way for next-generation precision targeted radionuclide therapy.
BetaBart

On October 3, 2024, Radiopharm Theranostics (an Australian-listed company, ASX: RAD) announced a significant advancement regarding BetaBart (RV-01), its B7-H3-targeting radiopharmaceutical.
The company has submitted a pre-IND meeting request to the FDA and successfully completed the production of the first GMP batch of the antibody and chelator.
BetaBart is the world's first radiopharmaceutical targeting the B7-H3 (4Ig isoform), developed in collaboration with MD Anderson Cancer Center. This specific target is overexpressed in various tumors and is associated with poor prognosis.

Preclinical data demonstrated that BetaBart effectively reduced tumor size and extended animal survival. The antibody has been engineered to shorten its blood half-life, thereby reducing off-target toxicity and enhancing its suitability for clinical applications.
The company plans to initiate a first-in-human Phase I/II clinical trial in mid-2025. This trial will utilize Lutetium-177 to treat various cancers. The isotope supply chain has been secured through multiple contracts.
Both the company's CEO and the research team from MD Anderson emphasized the significant outcomes of their collaboration and expressed strong anticipation for advancing the clinical trial.
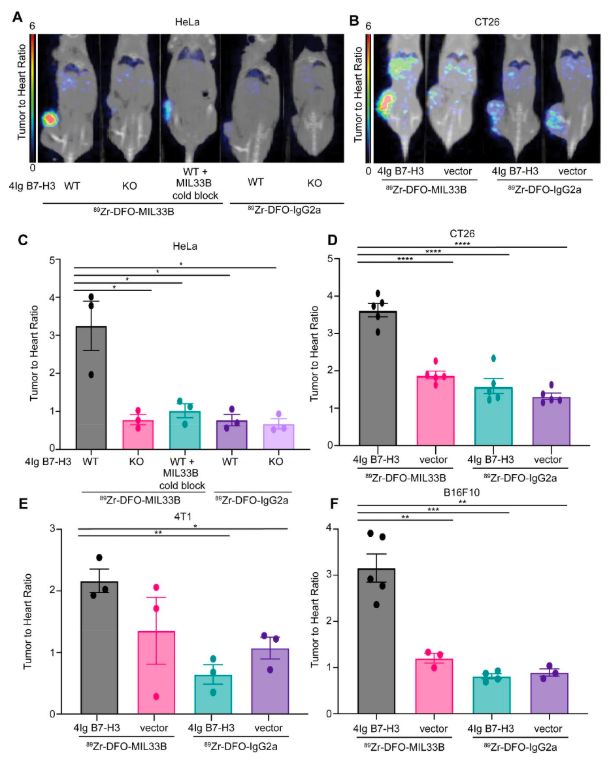
Analysis of the disclosed preclinical data—pertaining to the patent (licensed to Radiopharm Ventures) titled "Anti-Cancer Immune Priming with Beta-Radioligand Therapy and Isoform-Selective Targeting of 4Ig-B7-H3"—revealed that the anti-4Ig-B7-H3 monoclonal antibody, MIL33B, specifically targets the 4Ig-B7-H3 isoform on the tumor cell membrane (KD = 72 pM). This strategy effectively circumvents interference from soluble 2Ig-B7-H3.
Leveraging the "crossfire" effect of the beta-emitting radionuclide Yttrium-90, the therapy achieved a 53% long-term survival rate across multiple preclinical models.
Mechanistic studies unveiled that this treatment activates CD8+ T cell-dependent anti-tumor immunity through radiation-induced immunogenic cell death (ICD), thereby establishing immunological memory and overcoming the limitations of conventional radiotherapy which is restricted to local treatment.
Through live-cell screening, PET-CT target validation, and immunohistochemical analysis, the research confirmed the precise accumulation of MIL33B within the solid tumor microenvironment and its systemic immune activation effects.
Conclusion
In contrast to the rapid advancement of B7-H3-targeting ADCs, the development of radiopharmaceuticals for this target remains in its early exploratory stages. B7-H3-targeted RDCs, leveraging the radiation effects of beta or alpha particles, hold the potential to disrupt the current therapeutic landscape:
Furthermore, B7-H3-PET probes enable whole-body, dynamic visualization of target expression. This capability not only facilitates the identification of patient populations most likely to benefit from RDC therapy but also allows for real-time optimization of dosing regimens through dosimetry modeling, thereby advancing the paradigm of theranostics.