2025-10-27
Recently, exciting news has emerged in China's BNCT field: A research collaboration between Zhongbor Medical and Xiamen Humanity Hospital on the first application of BNCT in treating Mesenchymal Chondrosarcoma (MCS) has been officially published in "Advances in Radiation Oncology," an international authoritative journal sponsored by the American Society for Radiation Oncology (ASTRO). This represents not only the world's first successful exploration of BNCT technology in treating this rare cancer but also marks China's independently developed BNCT technology has achieved internationally leading clinical translational capabilities.

In this research, the domestically developed first-of-its-kind accelerator BNCT treatment system - NeuPEX (Extreme Frontier Knife), the specialized treatment planning software - NeuMANTA (Ray), and the domestically produced boron drugs (therapeutic drug BPA and companion diagnostic drug 18F-BPA), all independently developed by Boron Medical, were used to carry out treatment at the BNCT Center of Xiamen Humanity Hospital.
The results showed that after treatment, the tumor in the subject significantly shrunk, with the maximum diameter reduced by 45.8%, achieving a partial response (PR); during the long-term follow-up exceeding 500 days, the primary tumor continued to shrink and was effectively controlled, demonstrating excellent long-term local control efficacy.
From Despair to Rebirth: The BNCT Treatment Journey of the World's First MCS Patient
The subject participating in this study, a man in his 20s surnamed Dong (pseudonym), is not only China's first tumor patient to receive accelerator-based BNCT treatment but also the world's first patient successfully treated for mesenchymal chondrosarcoma (MCS) using BNCT technology. In 2022, Dong's life hit rock bottom: his facial chondrosarcoma recurred, and another surgery would risk disfigurement. After attempting various treatment methods including radiotherapy and chemotherapy, all failed. With the rapid progression of the tumor, eating and speaking became luxuries for the then 31-year-old Dong.
In October 2022, Adong received his first BNCT radiation therapy: "After the first treatment, I felt the facial tumor had shrunk by one-third, and I could gradually eat normally. I felt more energetic, like I was reborn." After the second treatment, the tumor on Adong's face had completely disappeared, allowing him to communicate and eat normally with others. His life gradually returned to normal. To this day, the tumor remains effectively controlled, his quality of life has continued to stay stable, truly achieving the leap from "treatment" to "normal life".
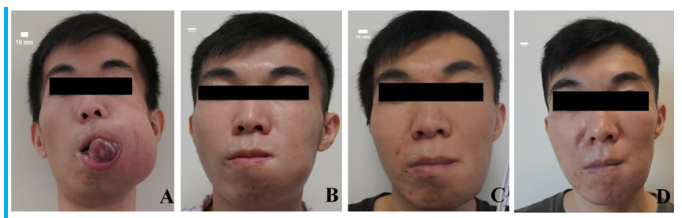
Significant improvement of tumor after BNCT: (A) Before BNCT treatment; (B) 90 days after the first BNCT; (C) 90 days after the second BNCT; (D) 150 days after the second BNCT
Professor Pan Jianji, the principal investigator (PI) of this clinical study, stated: "As the world's first successful case of BNCT treatment for chondrosarcoma, Adong's recovery has exceeded expectations. This not only demonstrates the unique advantages of BNCT technology in treating refractory tumors but, more importantly, relying on our domestic technological system, we have achieved precise and controllable treatment, providing valuable clinical evidence for future treatment of similar patients."
Behind the world's first treatment case lies the support of the entire domestic BNCT technology system constructed by Zhongbor Medical. Through the "high-end radiotherapy equipment + integrated diagnosis and treatment drugs + cloud-based treatment system", Zhongbor has formed a complete closed-loop of one-stop BNCT solution, providing a replicable innovative paradigm for global tumor treatment.
From launching China's first BNCT clinical research (IIT) in 2022, to entering BNCT drug-device combined registration clinical trials in 2024, and finally completing Phase I clinical patient enrollment in August 2025, the efficient progress made by Zhongbor Medical and Xiamen弘爱 Hospital not only serves as a vivid testament to China's high-end medical technology capabilities but also gradually transforms the original aspiration of "making BNCT accessible, affordable, and effective for every patient in need" from a vision into reality that benefits more patients.
Source: China Nuclear Technology Network